The presence of nanoparticles in plants, animals, and humans is on the increase and is rapidly becoming a major global health risk. There are many ways nanoparticles can enter the body, for example, inhalation, ingestion, skin exposure, and via medical procedures. The type and size of particles we are exposed to can vary but particles that are less than 2.5µm are a significant health concern. They can be breathed into the lungs and long-term exposure can result in increased mortality rates. Ultra-fine or nanoparticles, typically less than 100nm, can penetrate deep within the body and be widely distributed. Determining the location and the composition of nanoparticles within cells and tissues is crucial for understanding how they affect human health and what particles pose the greatest threat.
Airborne particles and air pollution are a major source of exposure and have been discussed in previous blogs and webinars, where energy dispersive x-ray spectrometry (EDS) provides an essential tool for the automated characterisation of particulate matter. External exposure to nanoparticles is not the only source of concern. Particles can also be generated internally from implant materials, wherever surfaces of different composition come into contact. Some types of implant generate more wear particles than others and can have a higher corresponding failure rate. Between 2003 and 2013, there was a very high incidence of hip implant failure in certain types of implant (between 5-37% within 5 years, according to the National Joint Registry), affecting just under 70,000 patients in the UK alone. It is not known why some patients reacted more than others, but tissues surrounding the implants obtained following joint failure showed high levels of wear particles.
Diagnostic tissue samples are often prepared using generic TEM sample preparation methods and are usually fixed, embedded in resin, and sectioned to a thickness of around 70-100nm. We can easily combine EDS with either TEM or SEM and detect the composition of any nanoparticles present in the sample, thus determining which parts of the implant they originated from.

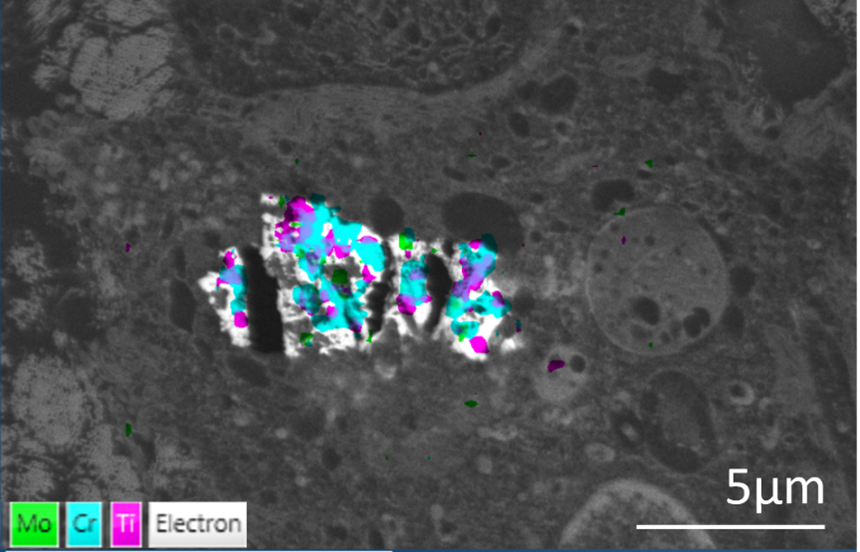
Two examples are shown here. Tissue samples were collected after the failure of hip implants and prepared for routine TEM examination. Thin sections of tissue were mounted onto copper grids and held using a STEM holder. EDS data was collected at 5kV in an SEM using an Ultim Extreme detector. The backscattered images showed clusters of nanoparticles within endosomes (arrow). EDS identified cobalt, titanium and chromium nanoparticles in the sample shown above, and particles containing chromium, titanium, and molybdenum in the other example. Even though these materials clearly originated from different regions on the hip implants, which were composite structures with several different types of material, they ended up being accumulated within the same cells. These particles could not be broken down and continued exposure to them resulted in large nanoparticle aggregates in cells (image below). This continued until toxic levels were reached, causing cell death.
EDS is a rapid analytical tool that provides compositional information in electron microscopes that can be used on samples prepared for routine EM analysis. In this case, EDS provided a diagnostic tool that helped identify problem areas on implants that will hopefully contribute to future implant design and improved patient outcomes.
EDS is not a technique normally considered applicable for biological samples, but above is only one of many different possible applications of EDS for life-sciences. For more information on biological applications, look at our on-demand webinar and our bioimaging and life science web pages.