Recent blog posts Learning to see more and More than just a comparison have highlighted some of the benefits of using a correlative analysis approach, combining data from more than one type of microscope to enhance our understanding of samples and interpretation of results. One of the ongoing challenges in doing any form of correlative microscopy is capturing images of the same region, especially when you may be using different scales of magnification and when the orientation of the sample may change between microscopes.
Our new Relate software focuses on the correlation of data collected using electron microscopes (EM) and atomic force microscopy (AFM). There are several issues that require consideration when planning a correlative experiment using these types of microscope and this will also depend to some degree on the type of sample being imaged. While AFM analyses can be conducted in liquid and air environments, samples are exposed to a vacuum in an EM and thus must be completely dry before being placed into the microscope. Samples being imaged with an electron beam often build up a fine layer of carbon on their surface during imaging, or they may require a some form of conductive coating before they can be imaged, and these layers may affect the AFM probe and any measurements you may take. Sample preparation and planning your experiments are key to successful correlative microscopy and it is useful to ask the following questions.
- Will you prepare the sample for both imaging environments before using either type of microscope or will you image with one first and then adapt the sample for the next imaging modality? How will this affect the sample?
- Which type of imaging will be used first on your sample and how might this affect the second microscope imaging conditions?
Once you have identified your sample preparation and order of imaging, you will have to identify what strategy you need for finding the same region on your sample. There are several methods that can be used.
Features as reference points
This is probably the most common and the easiest method to finding similar regions. Features will need to be visible at the lowest magnification scales that the microscopes are capable of and visible using both types of microscope. The features used can be part of the sample or defects/cracks within the sample that can be used as a guide. Be careful of using features that may move, such as dust particles that may accumulate on the surface of your sample.
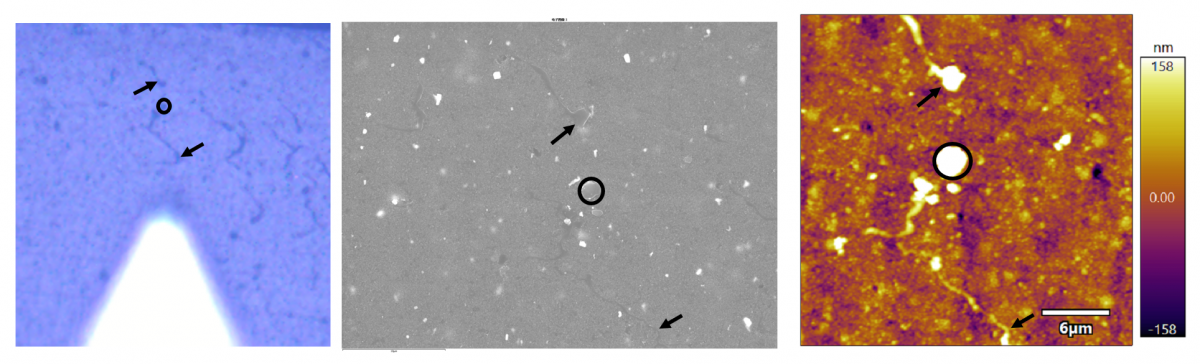
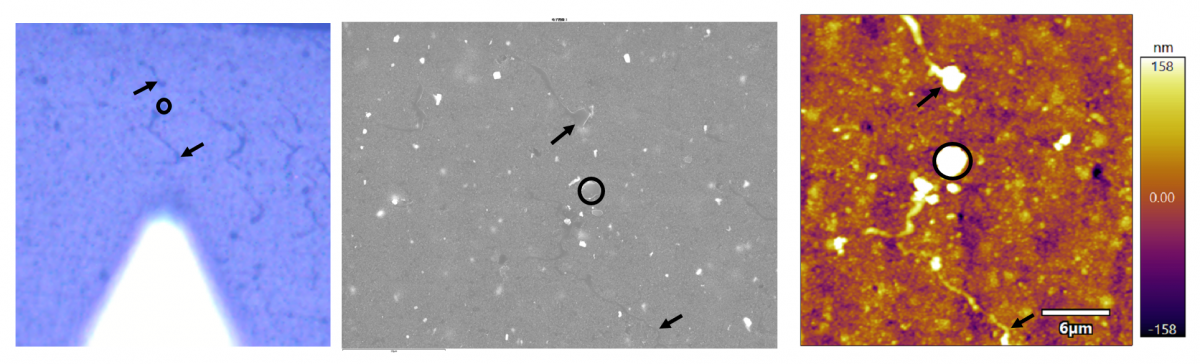
It is usually the case that sample-based features are going to be more obvious in one type of microscope than the other. An example is shown below where the lines that are obvious in the CCD image (left) are quite faint in the EM image (middle) and a bit more obvious in the AFM image (right).
Adding or creating fiducials
It may not always be possible to use features of your sample, especially when they are difficult to observe using one type of imaging. In this case, adding fiducial markers can help to find a specific area. Fiducial markers are objects placed onto or within the sample that are visible in both imaging modalities. Ideally the fiducial markers would be bound to the sample in some way so that they do not move when the sample is transported between microscopes. An example is the use of quantum dots in biological samples for correlative light and electron microscopy. If no obvious label is available, it may be possible to create fiducials by etching or depositing a pattern onto your sample, for example, by using a focused ion beam scanning electron microscope.
Guides and markers
Another way to help locating the region of interest between microscopes is to use some form of guide. Finder grids can be stuck to the surface of the sample or there are correlative microscopy EM stubs that the sample could be mounted onto.
The best solution will depend on your samples as well as availability of equipment, techniques, and materials. A combination of techniques might be the best solution, such as using a correlative SEM stub for finding your region of interest at lower magnifications, followed by using sample features when you get to higher resolution imaging.
Once you have collected your data you will need to analyse it. To learn more about our solution for analysis and visualisation of correlative data, take a look at our webpage on Relate or watch our short webinar which is available to watch on demand.